Methionine
Overview
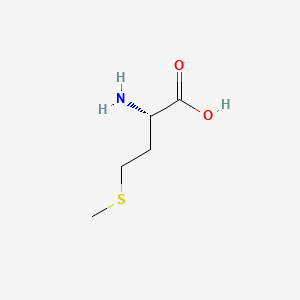
Methionine is an essential amino acid that plays a central role in methylation processes. It forms S-adenosylmethionine (SAMe), the universal methyl donor that fuels neurotransmitter synthesis (dopamine, norepinephrine, serotonin) and drives phospholipid methylation in neuronal membranes. Methionine is converted to SAMe through a process that involves B vitamins (B6, B2, folate, B12) as cofactors. After donating its methyl group, SAMe becomes S-adenosylhomocysteine, which is then converted to homocysteine, which can be remethylated back to methionine in a cycle that requires adequate B vitamin status.
Recipes
Foods
Biological Mechanisms and Implications
| Biological Target | Therapeutic Areas | Mechanism of Action |
|---|---|---|
| Methylation & One-Carbon Metabolism | Essential amino acid that forms S-adenosylmethionine (SAMe), the universal methyl donor for neurotransmitter synthesis and membrane phospholipid methylation |
References
- Methionine is essential and forms SAMe, the universal methyl donor for neurotransmitter synthesis and membrane phospholipid methylation
- B vitamins, particularly B6, B2, folate (5-MTHF), and B12, are essential cofactors in remethylation of homocysteine to methionine, which is converted to SAMe










